There is a big difference between “FDA approved” and “FDA cleared”. The FDA has 3 classifications for devices and medications; Class 1 (over the counter anti-snoring devices), Class 2 (dental implants, apnea devices, etc..) and Class 3 (heart valves, implanted parts, devices that are life sustaining and are complex devices that can be life threatening but if their benefits outweigh the risks) and the FDA will approve them if they
Class 1 and 2 medical devices are generally “cleared” by the FDA, which means the manufacturer demonstrated that their product is “substantially equivalent to another legally marketed device” that already has FDA clearance. The cleared products are called a predicate. We have embedded some applications and approvals so you can see how dental companies consistently reference others in their application and rely on approval of these predecessors. We have not found any dental devices, software, materials that fall into the Class 3 category
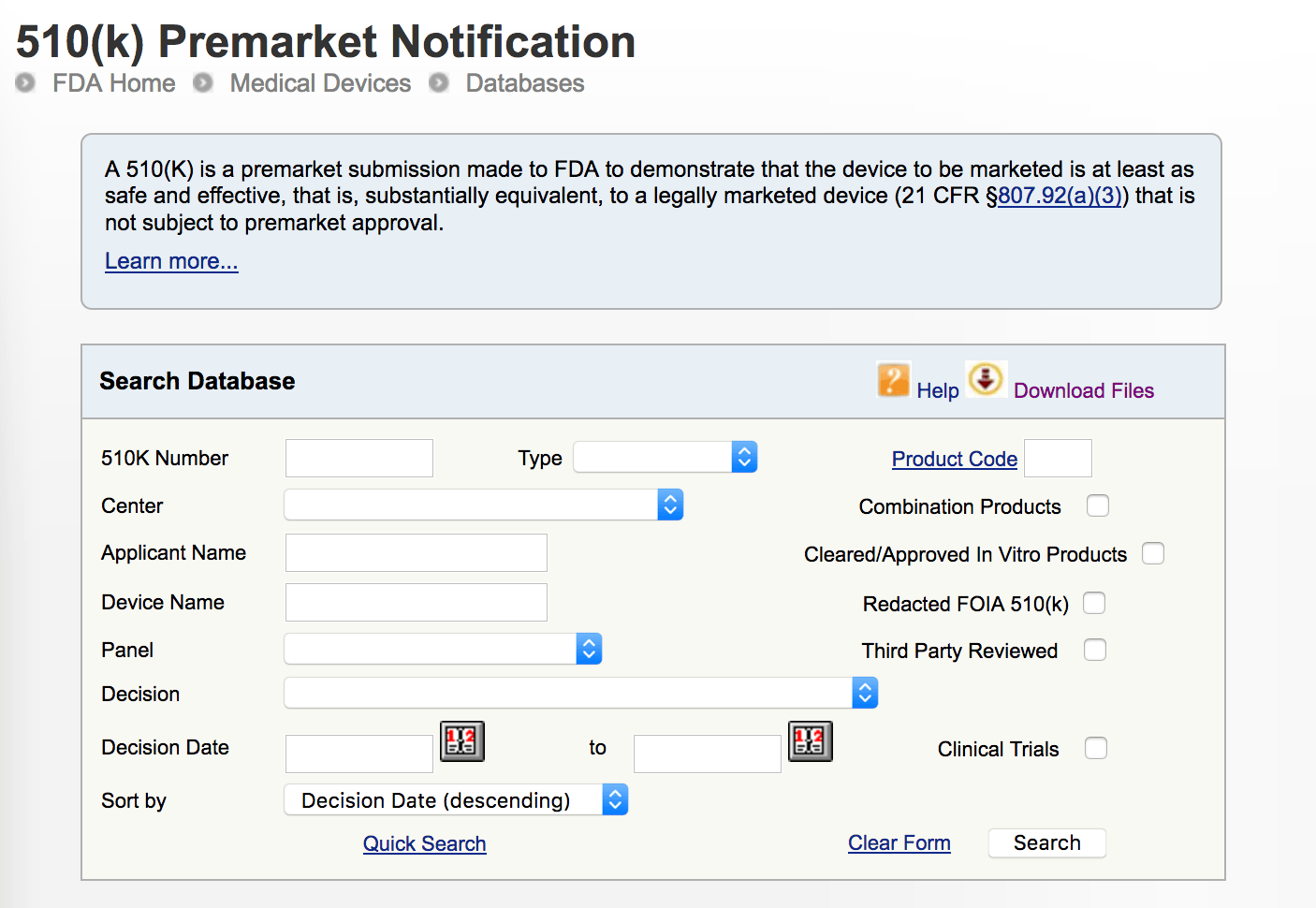
Note that “FDA cleared” does not validate a product or mean that a dental prosthesis will fit with great accuracy. You can click on the first image to read about the process in detail on the FDA website.
In the second image, you can search a device, manufacturer, or materials to see exactly how the application was submitted and the FDA’s response to the application
ABUTMENT SOFTWARE
3Shape Abutment Designer
K151455ORTHODONTIC SOFTWARE
CEREC Ortho Software
K1711223Shape Ortho Software
K152086IMPLANT SOFTWARE
Sicat 1.0
K090119Sicat 1.2
K103723Simplant
K0338493shape Implant Studio
K1415703Shape Implant Studio 2015
K152078APNEA DEVICES
Snore Hook

Optisleep
K153291







You must be logged in to post a comment.